
Now Enrolling: PrE0510 for Patients With Extensive-Stage Small Cell Lung Cancer
February 27, 2026
From the Co-Chairs, February 2026
February 27, 2026Now Enrolling: EA6232 Recurrence Prevention Study for Patients With a History of Melanoma
EA6232 - Sulforaphane for the Prevention of Melanoma in Patients with Multiple Atypical Nevi and a Prior History of Melanoma
Melanoma is one of the most common cancers in the United States, with diagnoses increasing more rapidly than most other common cancers despite public awareness campaigns recommending sunscreens and sun-protective behavior modification. Treatment for early-stage melanoma is generally highly successful, leading to 5-year survival rates of nearly 100% for localized cancer confined to the primary site, and 75% for cancer that has spread no further than the regional lymph nodes. More than 1.5 million people in the United States are living with melanoma (SEER Database).
The EA6232 clinical trial is for patients who have multiple atypical nevi (moles that can sometimes become cancerous) remaining after treatment of the primary site of melanoma. It explores a new prevention strategy for these patients, who are advised to use sun protection, perform self-exams, and visit their physician regularly for mole checks. Previous studies have shown that they are at substantially higher risk to develop a new melanoma compared to patients without multiple atypical nevi.
This phase 2 trial is exploring therapeutic prevention—using treatment to prevent, delay, or reverse the development of precancerous skin lesions into melanoma. It is evaluating sulforaphane, a natural antioxidant and anti-inflammatory substance found in cruciferous vegetables such as broccoli, cauliflower, and Brussels sprouts. Earlier research studies have shown that eating cruciferous vegetables is correlated with reduced risk of certain types of cancers, and that this benefit could be related to sulforaphane.
Sulforaphane is typically taken as a dietary supplement in pill form and has been shown to help neutralize toxins in the body and reduce chronic inflammation. Additionally, exposure to ultraviolet (UV) radiation is thought to be the leading cause of melanoma, and applying sulforaphane to the skin has been shown to protect against UV-related skin damage. Sulforaphane has also been shown to inhibit tumor growth and promote apoptosis (cell death) in cancer cells. Researchers are currently studying sulforaphane in clinical trials across multiple cancer types.
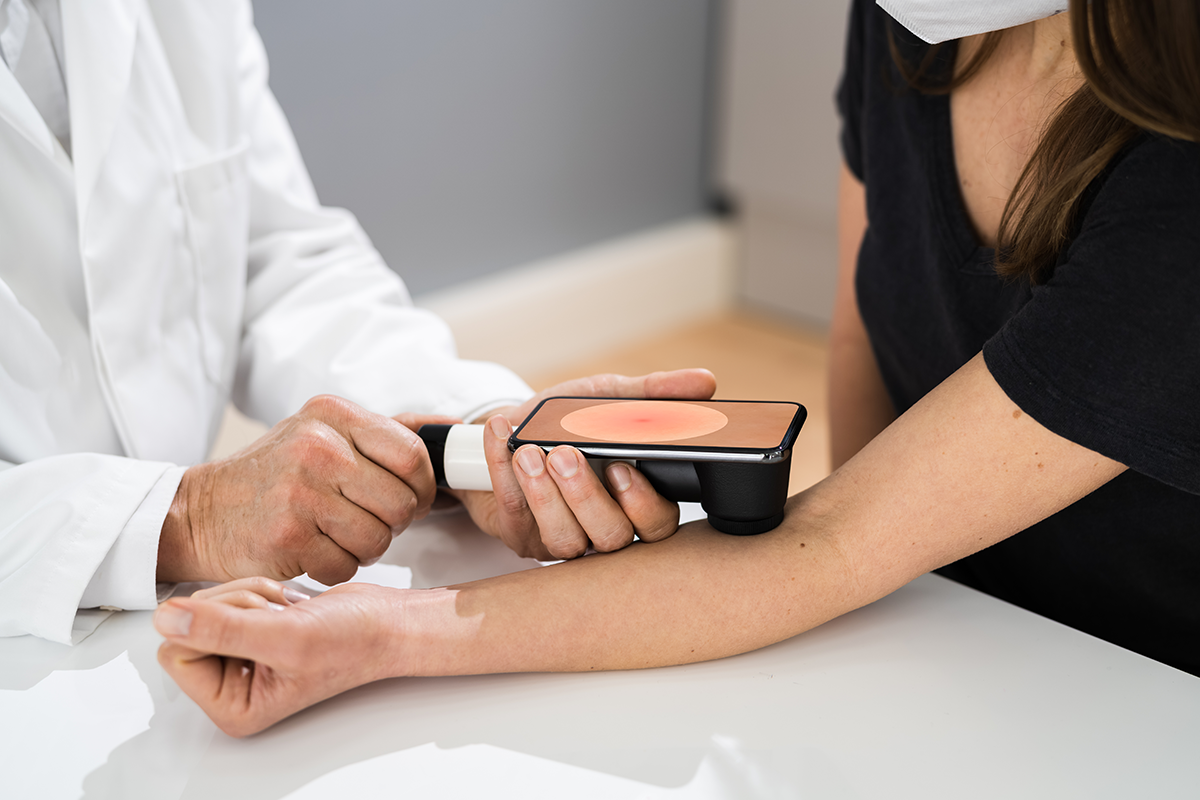
EA6232 builds on a successful phase 1 trial[i] and is testing whether daily oral sulforaphane (40 mg) can slow the growth of nevi and reduce the risk of melanoma recurrence. Researchers will compare changes in total nevus area over 12 months between patients receiving sulforaphane and those on placebo, with measurements taken at baseline, 3 months, and 12 months. Imaging assessments will utilize a publicly available software called DermaAI, which tracks moles and lesions over time for changes in size, number, and appearance. The hypothesis is that sulforaphane will limit increases in total nevus area and favorably modify cellular and molecular features in nevi and blood—reducing markers of inflammation and immune infiltration while increasing expression of downstream genes in the Nrf2 damage response pathway.
Patients are eligible for EA6232 if they were previously treated for early-stage melanoma, defined as either melanoma in situ, localized resected stage 1 or 2 node-negative melanoma, or resected node-positive stage 3 melanoma, and have at least 3 atypical nevi remaining. Eligible patients must not have received treatment for melanoma within the last 365 days or have an allergy to cruciferous vegetables.
Approximately 120 patients will take part in the study. They will be randomly assigned by a computer to receive either sulforaphane or placebo for one year, given as pills to be taken daily. Participants will then be followed by their doctor for another year afterward.
EA6232 offers patients and researchers an opportunity to contribute to melanoma recurrence prevention research using a low-risk substance. If successful, this study will identify a clinical signal that will justify a large national trial for the further development of sulforaphane in skin cancer prevention.
Learn more about the EA6232 study at ecog-acrin.org
The study chair for this trial is John M. Kirkwood, MD (University of Pittsburgh/UPMC Hillman Cancer Center). The dermatopathology co-chair is Ari Karunamurthy, MD (University of Pittsburgh/UPMC Hillman Cancer Center), and the community co-chair is Gary Cohen, MD (Consultant).
[i] Tahata S, Singh SV, Lin Y, Hahm ER, Beumer JH, Christner SM, et al. Evaluation of biodistribution of sulforaphane after administration of oral broccoli sprout extract in melanoma patients with multiple atypical nevi. Cancer Prev Res (Phila). 2018 Jul 1;11(7):429–38.
![ECOG-ACRIN logo[19516]275×75](https://blog-ecog-acrin.org/wp-content/uploads/2021/03/ECOG-ACRIN-logo19516275x75.png)
